ICSR and SAE / SUSAR Management
What is Pharmacovigilance?
Pharmacovigilance serves a fundamental purpose for the detection, assessment, understanding and prevention of adverse effects or any other harm associated with medicinal products.
What is Valid Individual Case Safety Reports (ICSR)?
A case reported with minimum four criteria (i) an identifiable reporter, (ii) an identifiable patient, (iii) a suspected drug and (iv) an adverse event, is considered as a valid case or Individual Case Safety Reports (ICSRs) and should be submitted to regulatory authority based on the day-zero, event seriousness and other applicable factors. Timeline for the submission of ICSR starts from the "Day zero", which is a date on which an organisation becomes aware of minimum four valid criteria for ICSR submission.
ICSR Reporting Requirement
The timeline for the submission of valid ICSRs is factored based on their source of reporting and event seriousness. All cases from solicited sources reported with Serious Adverse Event (SAE) should be submitted to regulatory authority within 07-calendar days, if reported event is fatal/life-threatening; and should be submitted to regulatory authority within 15-calendar days, if reported event is Suspected Unexpected Serious Adverse Reaction (SUSAR).
All cases from unsolicited sources reported with serious adverse event should be submitted to regulatory authority within 15-calendar days and all cases from unsolicited sources reported with non-serious adverse event should be submitted to regulatory authority within 30-calendar days or 90-calendar days or with the periodic reports as per local requirement. Actual timeline for ICSR submission to the regulatory authority may vary in different countries as per local requirement. If require, event expectedness should also be considered as one of the factors for expedite reporting as per local requirement (e.g., USA, Canada).
ICSRs requires submission to regulatory authority as an E2B(R2) or E2B(R3) compliant xml file, or CIOMS form, or MedWatch form, or any other ADR reporting form as per local requirement.
Lifecycle of ICSR for Complete Pharmacovigilance Activity
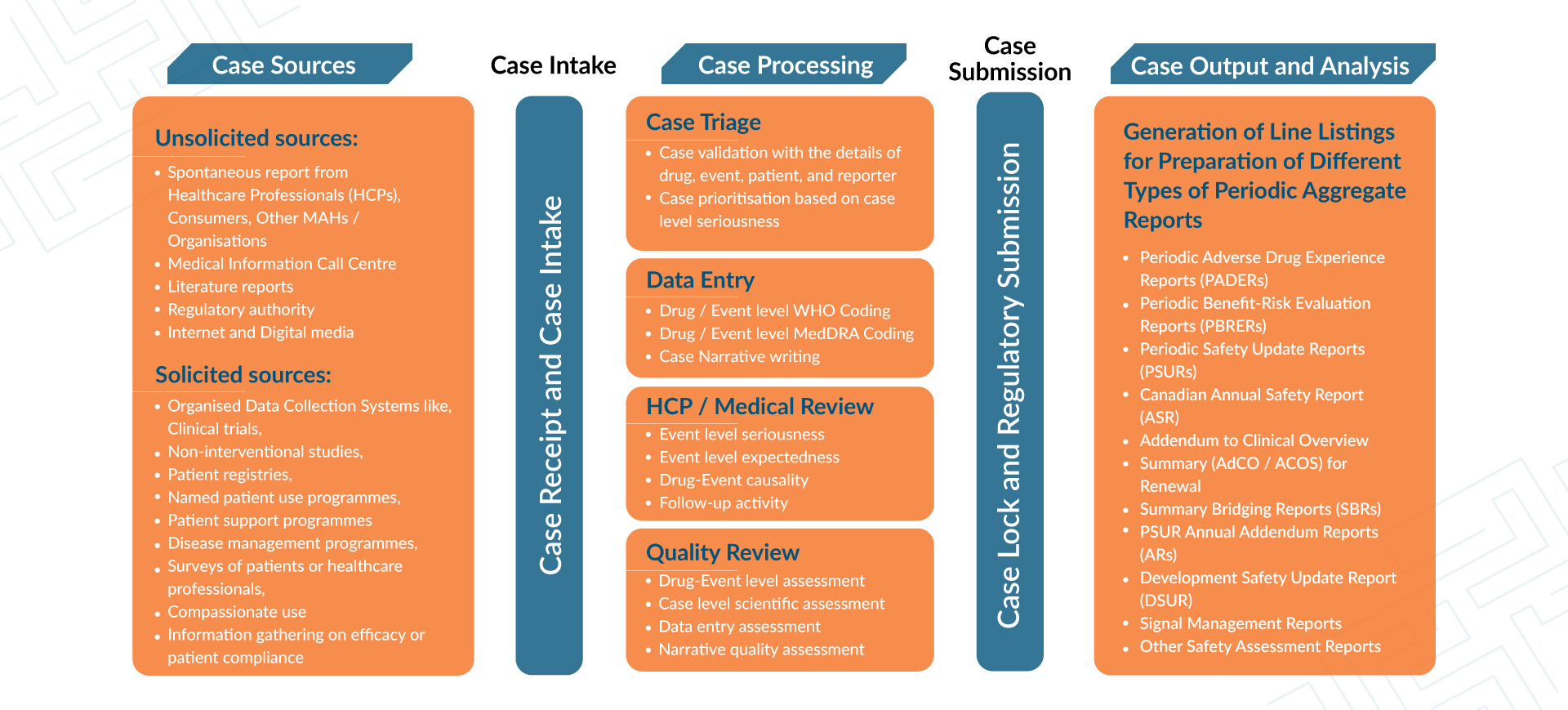
Data Access and Monthly Compliance
AllyWorth using the safety database which provides real-time access to all data, including adverse event or adverse drug reactions reports, ICSRs, and SAE / SUSARs in our system. We share the monthly compliance data with MAHs to provide a quick overview for all the cases, which are processed and submitted on time, ensuring the compliance with legal obligations.
Case Reconciliation
A robust process of case reconciliation ensures the exchange of all relevant safety information between different parties as defined in Safety Data Exchange Agreement or Pharmacovigilance Agreement or contract.
AllyWorth performs the MAH specific case reconciliation activities with following parties on periodic basis as per the executed agreement, and safety management plans.
- Company Affiliates
- Business Partners
- Medical Information and Call Centre Team
- Clinical Trials Team
Our Solution for Complete ICSR Activity
We prefer to provide complete ICSR management service via client hosting Global Safety Database as per client requirement. If requied, we also have a partner to offer the service for Global Safety Database (like ARGUS, ARISg, and other customised database)
Given the strict timeline in place for expedited reporting or periodic reporting of all ICSRs including SAE and SUSAR, it is essential to partner with a reliable service provider like AllyWorth to ensure the optimal regulatory compliance.






